Autre
Septembre 2006Future therapies for eradication of hepatitis C
Hepatitis C virus (HCV) is a significant cause of morbidity and mortality, infecting 170 million people worldwide. The current standard therapy of chronic hepatitis C is pegylated interferon alpha (PegIFN) combined with ribavirin, providing cure of the virus in about one half of naïve patients (1,2). However, treatment is costly, prolonged (6 to 12 months), not suitable for all patients, associated with significant morbidity, and requires a substantial commitment from patients and health care providers. Fortunately, many new treatments for HCV are currently being designed and evaluated, i.e. new interferons, ribavirin analogues and small molecule HCV enzyme inhibitors.
The current overview provides the most recent information in these fields as presented at the 40 th Annual EASL Meeting in Vienna, April 2006 and the International Symposium on Viral Hepatitis, Paris, July 2006.
- New interferonsAdding polyethylene glycol (PEG) to therapeutic interferon alpha molecules can dramatically increase plasma exposure following dosing and leads to increased response rates. A recombinant fusion molecule of interferon alpha-2b and human albumin (Albuferon, Human Genome Sciences) has entered clinical trials. An interim analysis of a phase IIb study investigating efficacy and safety of albuferon combined with ribavirin 1000-1200 mg qd, in genotype 1 chronic HCV IFN-naïve patients was presented. At 12 weeks HCV RNA was undetectable in 74.5% of patients (n = 110) treated with albuferon 1200 µg q2wk vs 65.8% (n = 114) in patients treated with peginterferon alpha-2a (3,4). Early viral response (HCV RNA reduction ≥ 2 log 10 at week 12) was 87.5% vs 85,7% the peginterferon alpha-2a arm. The data suggest that albuferon may offer efficacy and safety comparable to peginterferon alpha-2a with improved dosing schedule. A phase III study with albuferon q2wk is planned.
- ViramidineViramidine (Valeant Pharmaceuticals) is an inactive prodrug of ribavirin that is activated through deamination in the liver by adenosine deaminase (Fig. 1). Preferential viramidine delivery in the liver and poor uptake in erythrocytes results in reduced haemolytic anaemia. The results of a phase III study were presented (5,6). The study consisted of 970 patients randomized to receive viramidine 600 mg bid or weight-based ribavirin in combination with peginterferon alpha-2b. The rate of anaemia (Hb < 10 g/dL) was 24% in the ribavirin group vs only 5% in the viramidine group (p < 0.001). A sustained viral response (SVR) was obtained in 62% with ribavirin vs 52% with viramidine. Analysis of weight based dosing of viramidine demonstrated that increasing the mg/kg dose of viramidine improved the response without a proportional increase in anaemia (viramidine > 23 mg/kg SVR of 81% with anaemia in 12.5%).
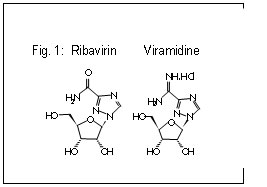
Although viramidine may not show an incremental higher virologic response compared with existing combination therapy, improvements in the safety profile over ribavirin would represent important clinical progress and would likely increase the proportion of patients currently eligible for therapy.
- HCV enzyme inhibitorsThe HCV genome encodes 4 enzymes. Several HCV enzyme inhibitors are in clinical trials. The 2 most advanced classes of agents are those directed at NS3 protease (Fig. 2) and NS5B RNA dependent RNA polymerase (Fig. 3).
- HCV specific protease inhibitorsThe NS3 region comprises a multifunctional protein that contains a serine protease at the N-terminus that cleaves the viral polyprotein into functional proteins essential for replication.
BILN-2061 (Boehringer-Ingelheim) was the first HCV protease inhibitor to enter in clinical trials. It demonstrated a transient but significant and rapid decline in HCV RNA levels in HCV infected patients. Further development of this compound was halted because of cardiotoxicity in laboratory animals.
Other serine protease inhibitors have advanced to the clinic.
VX-950 (Vertex Pharmaceuticals) is an oral specific NS3 protease inhibitor. In a phase Ib study, HCV genotype-1 infected patients were dosed for 2 weeks with 450 or 750 mg VX-950 q8h, 1250 mg q12h or placebo. The highest plasma levels were noted in the 750 mg dose group, with a median 4.4 log 10 reduction in HCV RNA levels (7).
A 2 week study was designed to explore the viral kinetics of VX-950 in combination with peginterferon alpha-2a (8). Eight patients received VX-950 (750 mg q8h) + PegIFN on d1 and d8; 8 patients received VX-950 alone and 4 pegIFN alone on d1 and d8. The drop in HCV RNA in the patients receiving PegIFN alone was 1 log 10; in the patients treated with VX-950 alone 4.0 log 10 (with rebound or plateau phase in 4/8), whereas in the combination group HCV RNA dropped 5.5 log 10, with HCV RNA < 10 IU/mL in 4/8). This study shows clearly the superiority of a combination of VX-950 and PegIFN. A phase II study is planned to evaluate whether VX-950 in combination with PegIFN may allow HCV eradication with significant shorter duration.
SCH 503034 (Schering-Plough) is an orally active protease inhibitor. In a phase II study of genotype 1 infected patients who were previous nonresponder to pegIFN, 450 mg SCH 503034 tid was associated with a mean HCV RNA reduction of 2 log 10 from baseline (9). Combination of SCH 503034 and peginterferon alpha-2b in HCV-1 peginterferon alpha-2b nonresponders provided greater antiviral activity than either drug as monotherapy (10). Its activity and safety are further evaluated in phase III studies assessing 24 and 48 weeks of combination treatment.
- Polymerase inhibitorsThe NS5B region of the HCV genome encodes a unique HCV RNA dependent RNA polymerase that is essential for HCV replication allowing for selective inhibitor development.
NM283 (Valopicitabine, Idenix Pharmaceuticals) (Fig. 4) is a nucleoside analogue that induced a 1.2 log 10 decline in HCV RNA in genotype 1 infected patients receiving 800 mg daily for 2 weeks (11). An ongoing phase IIb trial is evaluating NM283/pegIFN combination for 48 weeks in 174 treatment-naïve patients with HCV-1 (12). At week 8, 96% of patients reached ≥ 2 log 10 drop in HCV RNA in the combination group, with mean drop of 4.5 log 10, and PCR neg in 56%.
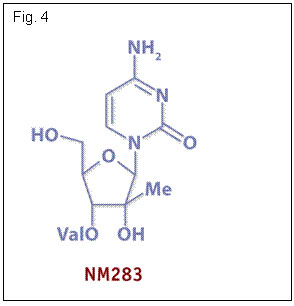
These data confirm the preclinical data suggesting synergy between interferon and NM283. NM283 is currently also being studied in a phase IIb trial of 190 HCV-1 infected patients failing early viral response on pegIFN/ribavirin (13). Patients are receiving NM283 alone, NM283 and peginterferon alpha-2a or peginterferon alpha-2a + ribavirin for 48 weeks. At 24 weeks HCV responses in the higher dose NM283 + pegIFN were significantly greater than in pegIFN/ribavirin retreatment (mean drop HCV RNA of 3.32 log 10). As the drop was substantially lower than in treatment-naïve patients (12) confirming the difficulty in suppressing HCV RNA in nonresponder patients.
R1626 (Roche) is a prodrug of the nucleoside analogue R1479, a potent inhibitor of the polymerase enzyme in vitro. Administration of R1626 1500 mg bid for 2 weeks in HCV infected, treatment naïve patients resulted in HCV RNA reduction of 1.2 log 10 from baseline (14). It will be further evaluated in phase II combination clinical studies.
- HCV specific protease inhibitorsThe NS3 region comprises a multifunctional protein that contains a serine protease at the N-terminus that cleaves the viral polyprotein into functional proteins essential for replication.
Conclusions
Many new drugs are currently being evaluated for improving current standard therapy of chronic hepatitis C. Protease inhibitors seem to be very potent in vivo in treatment-naïve and interferon-nonresponder patients, whereas polymerase inhibitors are less potent in monotherapy. Resistance seems a major drawback. Probably combination with immunomodulatory treatment with interferon-based therapy will be necessary to eradicate the virus. The results of ongoing phase II studies on this issue are eagerly awaited.
References
- Manns MP et al. Peginterferon alpha-2b plus ribavirin compared with interferon alpha-2b plus ribavirin for initial treatment of chronic hepatitis C: A randomised trial. Lancet 2001; 358: 958-965
- Fried MW et al. Peginterferon alfa-2a plus ribavirin for chronic hepatitis C virus infection. N Engl J Med 2002; 347: 975-982
- Benhamou Y et al. Phase 2b interim (week 12) results of albumin interferon alpha-2b combined with ribavirin in IFN-naïve, genotype 1 chronic hepatitis C infection. J Clin Virol 2006; 36: S38
- Zeuzem S et al. Interim (week 12) phase 2b virological efficacy and safety results of albumin interferon alpha-2b combined with ribavirin in genotype 1 chronic hepatitis C. J Hepatol 2006; 44 (Suppl 2): S270
- Benhamou Y et al. The safety and efficacy of Viramidine® plus pegylated interferon alpha-2b versus ribavirin plus pegylated interferon alpha-2b in therapy-naïve patients infected with hCV: phase 3 results. J Hepatol 2006; 44 (Suppl 2): S273
- Murphy B. The safety and efficacy of Viramidine® plus pegylated interferon alpha-2b versus ribavirin plus pegylated interferon alpha-2b in therapy-naïve patients infected with hCV: phase 3 results. J Clin Virol 2006; 36: S19
- Reesink HW et al. Final results of a phase 1B, multiple-dose study of VX-950, a hepatitis C virus protease inhibitor. Hepatology 2005; 42: 234A
- Reesink HW et al. Initial results of a 14-day study of the hepatitis C virus inhibitor protease VX-950, in combination with peginterferon-alpha-2a. J Hepatol 2006; 44 (Suppl 2): S272
- Zeuzem S. et al. Anti-viral activity of SCH 503034, a HCV protease inhibitor, administered as monotherapy in hepatitis C genotype-1 (HCV-1) patients refractory to pegylated interferon (PEG-IFN-α). Hepatology 2005; 42: 233A
- Zeuzem S. et al. The HCV NS3 protease inhibitor SCH 503034 in combination with Peg-IFNα-2b in the treatment of HCV-1 Peg-IFNα-2b non-responders: antiviral activity and HCV variant analysis. J Hepatol 2006; 44 (Suppl 2): S35
- Afdhal N et al. Final phase I/II trial results for NM283, a new polymerase inhibitor for hepatitis C: antiviral efficacy and tolerance in patients with HCV-1 infection, including previous interferon failures. Hepatology 2004; 40: 726A
- Dieterich D. Early clearance of HCV RNA with valopicitabine (NM283) plus Peg-interferon in treatment-naïve patients with HCV-1 infection: first results from a phase IIb trial. J Hepatol 2006; 44 (Suppl 2): S271-2
- Afdhal N. et al. Valopicitabine (NM283), alone or with Peginterferon, compared to Peginterferon/ribavirin (PEGIFN/RBV) retreatment in hepatitis C patients with prior non-response to PEGIFN/RBV: week 24 results. J Hepatol 2006; 44 (Suppl 2): S19
- Roberts S et al. Interim results of a multiple ascending dose study of R1626, a novel nucleoside analog targeting HCV polymerase in chronic HCV patients. J Hepatol 2006; 44 (Suppl 2): S26

